COVID-19, Year 2: 📈 Mixed news out of South Africa; US cases continue steady decline : Sunday 07 February 2021
Democrats move quickly on $1.9 trillion COVID relief bill; efficacy explainer; SuperBowl Sunday or #SuperSpreaderBowl?
Despite the welcome decline in daily cases being reported in the U.S., we’re still almost two times the summer peak (67,613, seven-day average).
My bet: in about five days, the case rate will begin rising again. Perhaps, if the case decline is a result of 41 million Americans having gotten at least one shot of a two-dose vaccine, the incline will not be as steep as it was during the fall. This is not a pipe dream: Oxford University research published last week suggests that a single dose of that vaccine protects against transmission of the virus.
However, deaths remain stubbornly stuck at about 3,000 per day (seven-day average). Deaths have not trended down with cases as they did in the prior four surges.
At 5:30 am Friday, after the Senate pulled an all-nighter, the Biden Administration moved forward on a COVID-19 relief bill that no one should be calling a “stimulus.”
“I’ve told both Republicans and Democrats, that’s my preference, to work together,” Biden told reporters on Friday. “But if I have to choose between getting help right now to Americans who are hurting so badly and getting bogged down in a lengthy negotiation or compromising on a bill that’s up to the crisis, that’s an easy choice. I’m going to help the American people who are hurting now.”
This is social insurance that is more akin to a response to the Great Depression than bailing out Wall Street in 2007-2008. Or the savings & loan industry in 1989.
The world has “allowed the virus to infect 100 million people already. That is 100 million chances for mutation,” virologist David Ho, who heads the Aaron Diamond AIDS Research Center at Columbia University, told Bloomberg.
What he failed to note is that the United States, with about 4% of the world’s population, accounts for 1-in-4 of those infections, a rate so disproportionate to our place on the world stage that I rarely see it mentioned in any domestic news article about COVID-19.
Thus the disastrous policies of the Trump Administration have provided rampant opportunity for SARS-CoV-2 to mutate, from the UK (B117) variant to those from South Africa (B1351) and Brazil (P1 and P2).


Data from a trial in South Africa of the Novavax vaccine suggest that reinfection is a real possibility as the virus (naturally) mutates. The South African variant, B.1.351, had become dominant during the vaccine trial period. People in the placebo arm of the trial who had antibodies (sign of a prior infection) got sick at the same rate (about 4%) as those who had no history of infection.
[I]t would not be surprising to see infection in people who are vaccinated, especially a few months out from vaccine,” said Michel Nussenzweig, head of the Laboratory of Molecular Immunology at Rockefeller University. “The key is not whether people get reinfected, it’s whether they get sick enough to be hospitalized.”
[…]
If it turns out that previously infected people could be susceptible to reinfection by variants, that could have implications for when the nation reaches herd immunity.
Cue Thomas Frieden, former director of the U.S. Centers for Disease Control and Prevention, told the WSJ that “we need to come to the acceptance phase that our lives are not going to be the same. I don’t think the world has really absorbed the fact that these are long-term changes.”
07 February 2021: what does efficacy mean?
Reporters are dropping epidemiological jargon into news stories whether or not it’s relevant; whether or not it’s accurate; and whether or not the terms have been explained.
Take the word efficacy. The “common” (dictionary”) meaning is “the ability to produce a desired or intended result.” And effectiveness: “the degree to which something is successful in producing a desired result; success.”
Vaccine efficacy is the reduction in disease risk among vaccinated persons relative to unvaccinated persons relative to the overall risk of disease (unvaccinated group risk) - all in an optimal (not real world) setting.
In this context, what does it mean that the BioNTech-Pfizer vaccine efficacy is 95%?
There were 43,448 adults in phase II/III trials from July to November 2020. Using random assignment, 21 ,720 people received two vaccine doses 21 days apart and 21 ,728 received two placebo shots.
At least seven days after the second dose, eight of those who had been vaccinated were diagnosed with COVID-19 (0.04%) compared to 162 who had received the placebo (0.75%). The NY Times provides a step-by-step lesson to calculate efficacy:
Thus, in the trial, the vaccine reduced infection risk by 0.71 percentage points. Dividing the difference in the two arms (0.71%) by the infection risk of the placebo group (0.75%) yields ~95% efficacy.
It does not mean that 95 out of 100 people will not get sick, which is what it sounds like.
The US already has 27 million reported cases: that’s more than an order of magnitude greater than projected from the trial results. One researcher estimates that one-third of the country has been infected, based on modeling.
That’s because 95% efficacy “means a 95% reduction in new cases of the disease in the vaccine group compared with the placebo group” under optimal conditions.
First, the trial group wasn’t just US citizens; it also included people from Argentina, Brazil, Germany, South Africa, and Turkey. Second, those “optimal conditions” means no prisons or nursing homes: places where people are crowded together and have gotten sick at rates greater than the general population.
To contrast efficacy with effectiveness, we (will eventually) look at how well the vaccine prevents “outcomes of interest” in a non-clinical (real-world) setting. Our outcome of interest is minimizing hospitalizations and death.
Many factors affect effectiveness: patient age, pre-existing conditions, time since vaccination, and “match to circulating strains.” That’s why personal protective gear and physical distancing will remain with us for an unknown length of time.
Finally, this cannot be stated too often: we do not know how effective COVID-19 vaccines will be at preventing infection and transmission (although we got a glimmer of hope this week). We do know that their primary aim is to save lives by reducing disease severity.
The SuperBowl #SuperSpreaderBowl




Shorts
Late Thursday, Johnson & Johnson submitted its request for emergency use authorization (EUA) for its COVID-19 vaccine candidate. The FDA Vaccines and Related Biological Products Advisory Committee will meet 26 February 2021 to vote on the EUA.
Mississippi has one of the worst racial gaps in vaccination rates in the country, even as Black residents are hit hardest by the pandemic.
Two shots of the Novavax vaccine and one from Johnson & Johnson (single dose) provide some protection from the South African variant. Although early trials suggested the Oxford-AstraZeneca vaccine provided limited protection against mild disease caused B1351, the country has halted its use.


Where are we today (07 Feb 2021)?
Summary, Johns Hopkins
US: 25,293,201 cases; 420,972 deaths
Global: 99,706,375 cases; 2,139,803 deaths
Vaccine report, CDC 💉
Doses distributed to states: 59,307,800
Total vaccinated: 9,147,185
People who have one shot of two: 32,063,752
Context
16 September 2020: “We’ll be able to distribute 100 million doses by the end of 2020 and a large number much sooner than that.”
08 October 2020: “We may have up to 100 million doses by the end of the year, enough to cover especially vulnerable population.”
15 December 2020: “We expect to have immunized 20 million of our American people” during the month of December. The Administration also pledged to distribute at least 40 million doses.
Vaccine map, Bloomberg
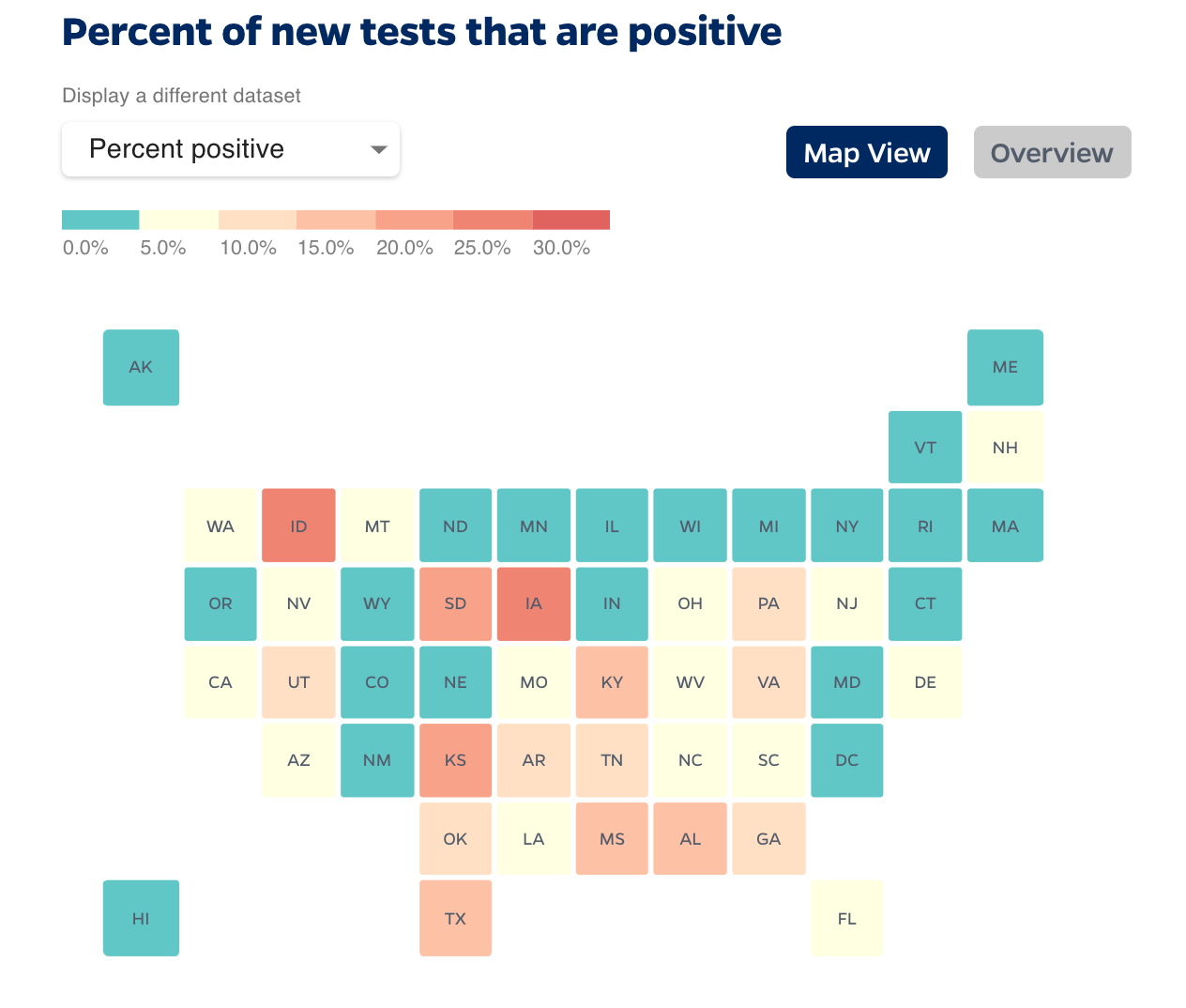
US Positivity rate, Johns Hopkins
On May 12, 2020, the World Health Organization advised governments positivity rates for testing should remain at 5% or lower for at least 14 days before reopening the economy. The Trump White House Task Force set a goal of 10%.
👓 See COVID-19 resource collection at WiredPen / See all newsletters